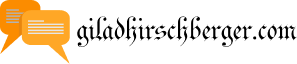You breathe air made up of atoms, eat meals composed of atoms and drink water made up of atoms-with a body that itself consists of atoms. Atoms move quicker when they’re of their gasoline kind (as a result of they are free to maneuver) than they do in liquid kind and solid matter. The weather are organized within the periodic desk based on the atomic quantity. We now know that atoms are made up of three particles: protons, neutrons and electrons — that are composed of even smaller particles comparable to quarks.
All atoms are made from the same bits, that are referred to as subatomic particles (“sub” means smaller than and these are particles smaller than atoms). Electrons are by far the smallest of the three atomic particles, their mass and measurement is too small to be measured using current know-how.
The potential energy of an electron in an atom is unfavorable , its dependence of its place reaches the minimal (probably the most absolute value ) contained in the nucleus, and vanishes when the distance from the nucleus goes to infinity , roughly in an inverse proportion to the space.
Earlier than we talk about the construction of atoms, we need to study the phrases mass and weight. The protons and neutrons cluster collectively in the central part of the atom, known as the nucleus , and the electrons ‘orbit’ the nucleus. To complicate things a bit more, we typically find atoms of a chemical ingredient that are a bit completely different to what we expect.
The so-referred to as “sturdy nuclear pressure,” the most highly effective drive that has but been described by the legal guidelines of physics, holds the atomic nuclei collectively. By rearranging these atoms and molecules you can stop something from manifesting or make it manifest.
Say electrons got here in a thousand variations of mass and electrical charge; ditto the other elementary particles. Moreover, carbon and hydrogen, there may be oxygen, nitrogen, sulfur, phosphorus and the halogens. They embrace carbon atoms joined in covalent bonds (electron sharing) with different atoms akin to hydrogen, oxygen, nitrogen, fluorine, chlorine, silicon, and sulfur.
Something that has mass and takes up space is a matter. Each atom consists of a nucleus and electrons that revolve around the nucleus at a substantial distance away. In electrically neutral atoms, the variety of protons equals the variety of electrons. The variety of protons in an atom is referred to as the atomic variety of that element.
The number of neutrons in relation to protons defines whether or not the nucleus is stable or goes by radioactive decay When there are too many neutrons or protons, the atom tries to make the numbers the identical by getting rid of the extra particles.
They usually contain metals and nonmetals as elements lively in ionic bonds are principally from reverse ends of the periodic desk with an electronegativity distinction exceeding 1.67. Being very robust, ionic bonds in compounds enhance melting factors and take a strong type in normal situations.
Beta decay happens when an atom has both too many protons, or too many neutrons. Matter waves are waves associated with transferring microscopic particles. As a matter of truth he prompt these particles have been subatomic components of the atom. This quantity is known as the atomic number.
Matter, Parts, And Atoms Khan Academy
The chemical properties of a substance (in other phrases what other atoms will it bond with, if it is liquid at room temperature, whether it is flammable, and so on) are primarily decided by the electrons on the outermost vitality level of the atom. Nuclear fission is when the nucleus splits into two smaller nuclei, releasing a lot of saved power This release of power is what makes nuclear fission useful for making bombs and electrical energy , within the form of nuclear energy The opposite method nuclei can change is through nuclear fusion , when two nuclei be part of together, or fuse, to make a heavier nucleus.
He called these particles “atoms”, which in Greek means “uncuttable”. There are 2,000,000,000,000,000,000,000 (that is 2 sextillion) atoms of oxygen in one drop of water—and twice as many atoms of hydrogen. If we examine an atom with a Kiwi fruit, the concentrated center portion might be compared to the positively charged atomic nucleus and the black color seeds to the negatively charged electrons.
Ozone Layer
Atoms are very small, but the exact dimension will depend on the aspect. In the event you join two different chemical elements collectively, you possibly can typically make a completely new substance.
Molecular Mass of a substance is the sum of the atomic masses of all atoms current in the MF of a substance or molecule. All carbon atoms have six protons. He said that matter was product of tiny indivisible particles called atoms and atoms of the same aspect had been all alike. Electrons encompass the atomic nucleus in pathways referred to as orbitals. Atoms aren’t just packets of matter: they comprise electrical energy too.
Each electron, or positron, will be identical in dimension, mass and electrical charge to as many decimal places as you care to measure. In 1803, English thinker John Dalton urged that parts had been tiny, strong balls made of atoms. Atoms are basic constructing blocks of matter, and cannot be chemically subdivided by odd means.
The Particle Adventure : Among the finest easy websites explaining atoms and the world inside them. Due to this fact, we are able to understand how the only molecule (bonding between two or more atoms) could be reworked into extra complex kinds in the presence of sunshine, temperature, and pressure.
Producing the lifeless out of the residing may confer with power (useless) rising from matter (dwelling), because the atom is in movement. If this drive had any extra binding energy, the protons and neutrons would mix with each other, and had been it any much less, they’d all spin away and the atom would disintegrate.
Balancing Of Chemical Equation
Atoms are all the pieces, in all places, anything, even what we consider nothingness is atoms, made up of atoms. Now buy a packet of three electrons (or their antimatter equal, the positron). This example demonstrates the gain, loss and sharing of electrons by atoms for possession of the same number of electrons as the noble gasoline in closest proximity on the periodic desk.
Suppose you make a list of the chemical parts so as of their atomic quantity (how many protons they’ve), beginning with hydrogen (H). 1803: English scientist John Dalton (1766-1844) printed the atomic idea of matter. The loss of electrons converts an atom into a positively charged ion, while the acquire of electrons converts an atom into a negatively charged ion.
Physics Articles
Our conception of the atom has undergone many changes because the day the idea that matter consisted of indivisible particles was first floated by the Indians and Greeks. 10 They’ve a unfavorable charge Protons and neutrons are of comparable measurement and weight to each other, 10 protons are positively charged and neutrons haven’t any charge. What number of bonds that fluorine will kind is determined by whether or not fluorine will gain or lose electrons.
Thomson, the British physicist who found the electron in 1897, proved that atoms actually could be divided, in response to the Chemical Heritage Basis He was able to decide the existence of the negatively charged particles by learning properties of electrical discharge in cathode-ray tubes.
atoms for peace ingenue lyrics
Everyone knows that the Atomic Theory was developed by an English chemist and physicist John Dalton. If an atom requires two electrons to obtain a noble gas configuration then it will acquire two electrons by sharing electrons (covalent) or by taking electrons (ionic). Chemists have lengthy known that once we mix totally different elements in chemical reactions, the components take part simple ratios.